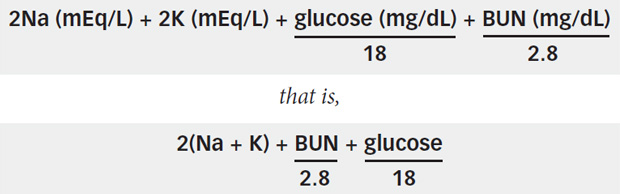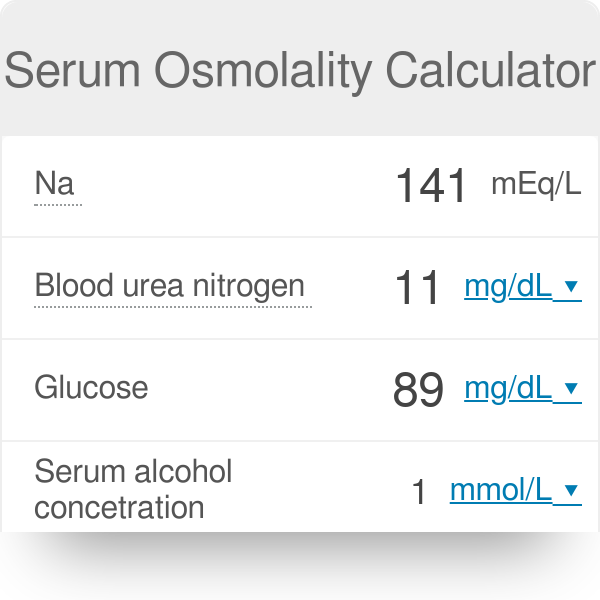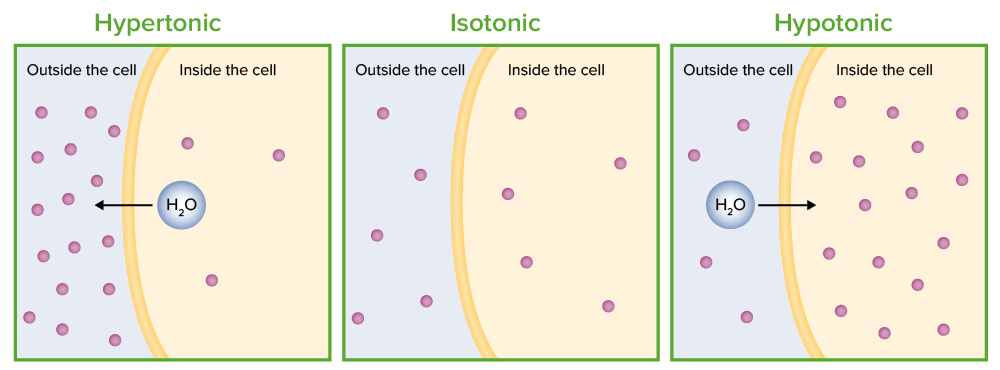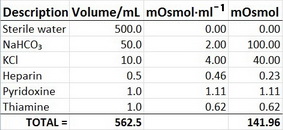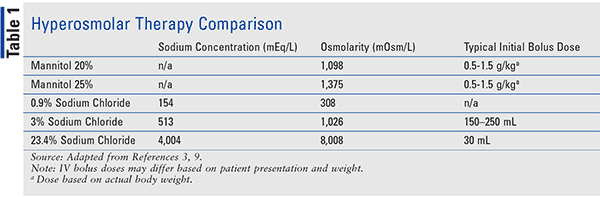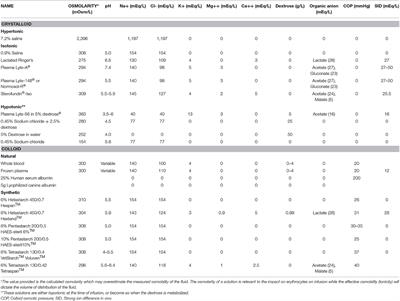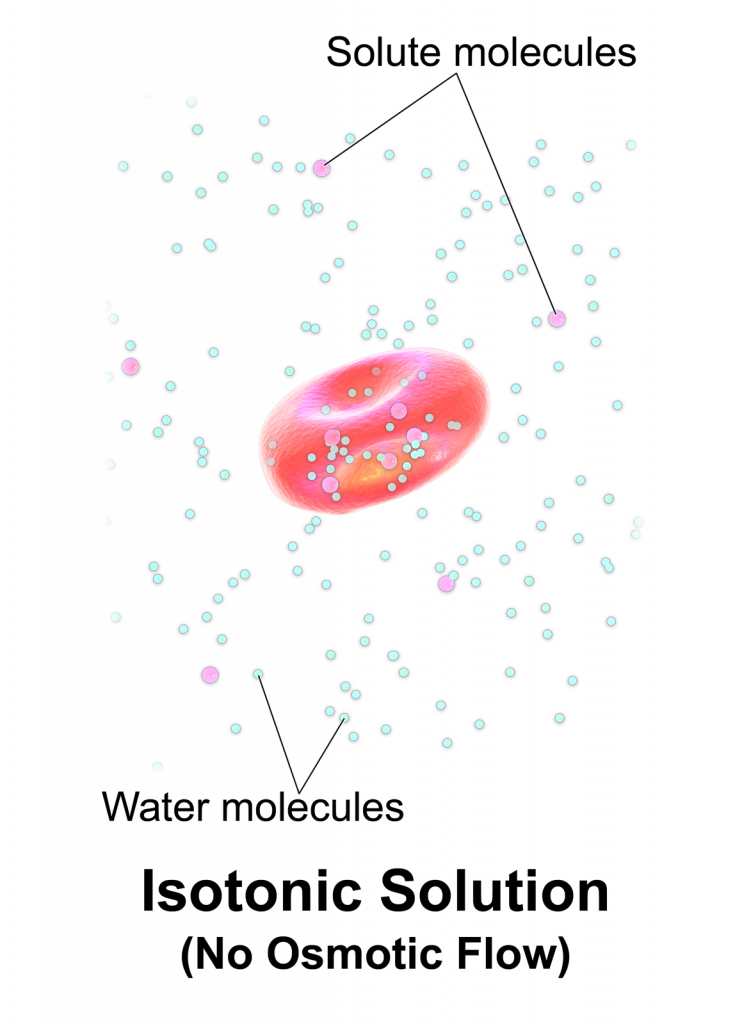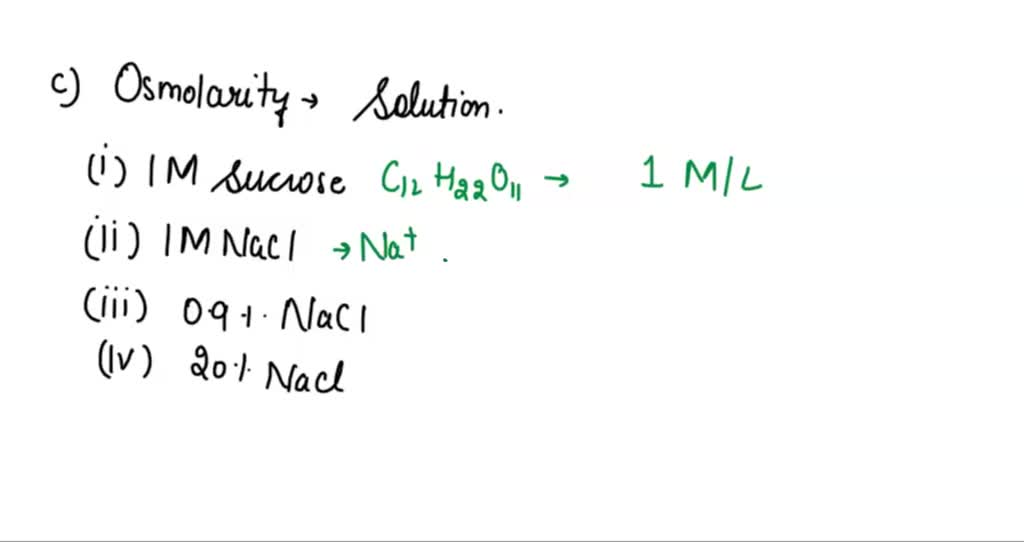
SOLVED: referred physiological isotonic sline solution (osmolarity 0.30 osmol) because it has A0.890 wlv NaCl solution ike same concentration of salts normal human blood; Calculate the osmolarity for the following solution and

Serum Osmolality Calculation & Overview | Serum Osmolality Definition - Video & Lesson Transcript | Study.com